Key Takeaways
- A novel computational model using differential equations can effectively simulate the complex interactions between cells, proteins, and chemokines during the proliferative phase of burn wound healing.
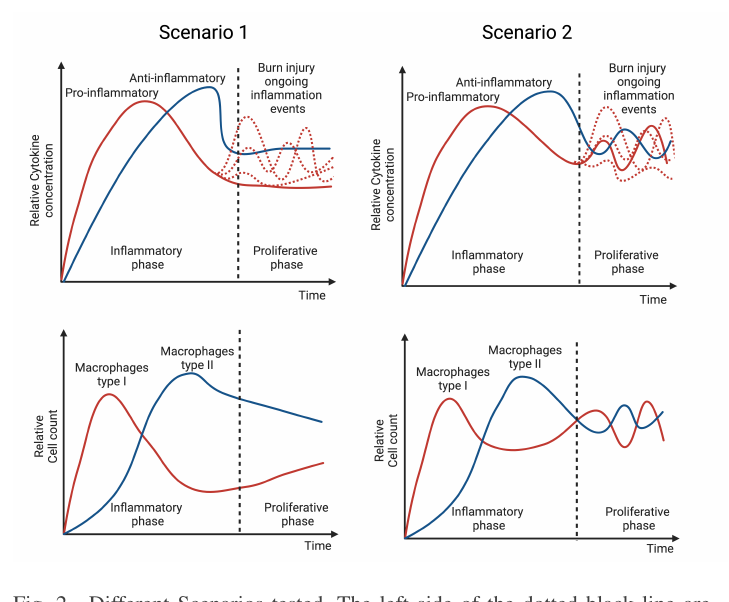
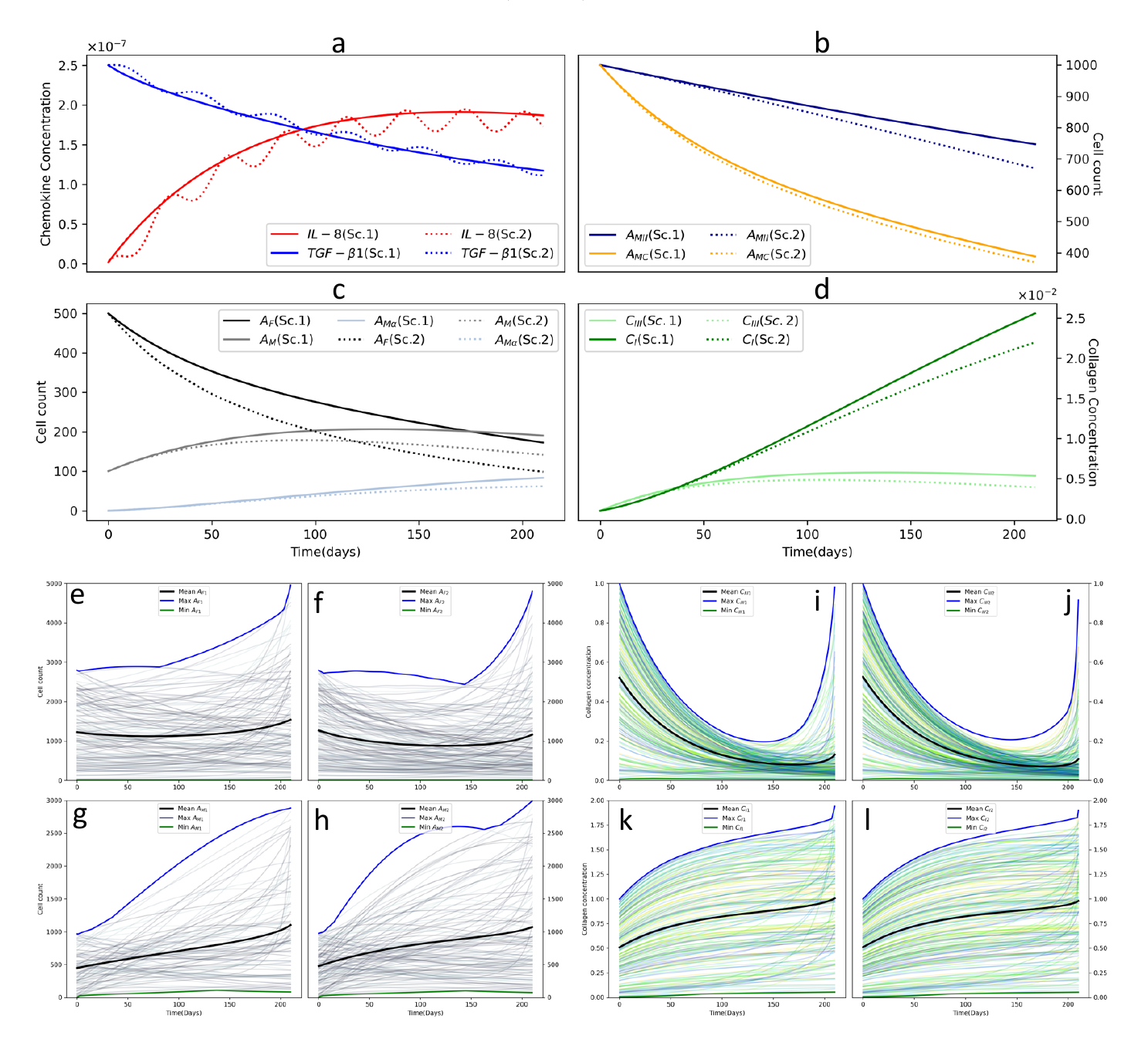
- The nature of inflammatory signaling (e.g., sustained versus fluctuating) is a critical factor that dictates the transition from the inflammatory phase to the proliferative phase, significantly impacting overall healing outcomes.
- The model highlights the crucial roles of fibroblasts and different collagen types in tissue repair, showing how their dynamic concentrations drive wound contraction and long-term tissue remodeling.
TL;DR
Burn wound healing is a complex biological process, and the proliferative phase is a critical step for tissue regeneration. However, the intricate interactions between inflammatory signals, cells like fibroblasts, and structural proteins like collagen are not fully understood. This makes it difficult to predict why some burns heal well while others lead to complications like scarring. Current research struggles to capture the long-term dynamics that determine the final healing outcome. This study introduces a computational model using differential equations to simulate the key cellular and molecular players in wound healing. By testing different scenarios of inflammatory signaling, the model demonstrates how these signals steer the healing process. The model successfully predicts the behavior of critical cells and proteins over time, showing its ability to simulate various healing outcomes, such as delayed repair or effective tissue remodeling, and offering a powerful new tool for burn research.
Why Does It Matter?
This paper provides a powerful in silico framework for studying the complex dynamics of burn wound healing. It enables researchers to test hypotheses about cellular and molecular interactions virtually, potentially accelerating the development of new therapies. By bridging the gap between biological observation and computational prediction, the model helps pave the way for more personalized treatment strategies in wound care.
Decoding Burns
Decoding Burns perfectly encapsulates this paper's core mission: translating the complex biological 'code' of wound healing into a predictive mathematical framework. The study utilizes a sophisticated computational model to decipher the intricate dynamics between key cellular players (macrophages, fibroblasts) and signaling molecules. This novel approach allows for the simulation of different healing trajectories, revealing how the initial balance between pro- and anti-inflammatory signals critically determines the ultimate outcome. By modeling these complex cellular interactions, the research demonstrates how a burn's progression towards either successful tissue remodeling or problematic fibrosis can be understood and potentially forecasted. The key insight is that such decoding tools enable a shift towards predictive medicine, paving the way for personalized interventions in burn care.
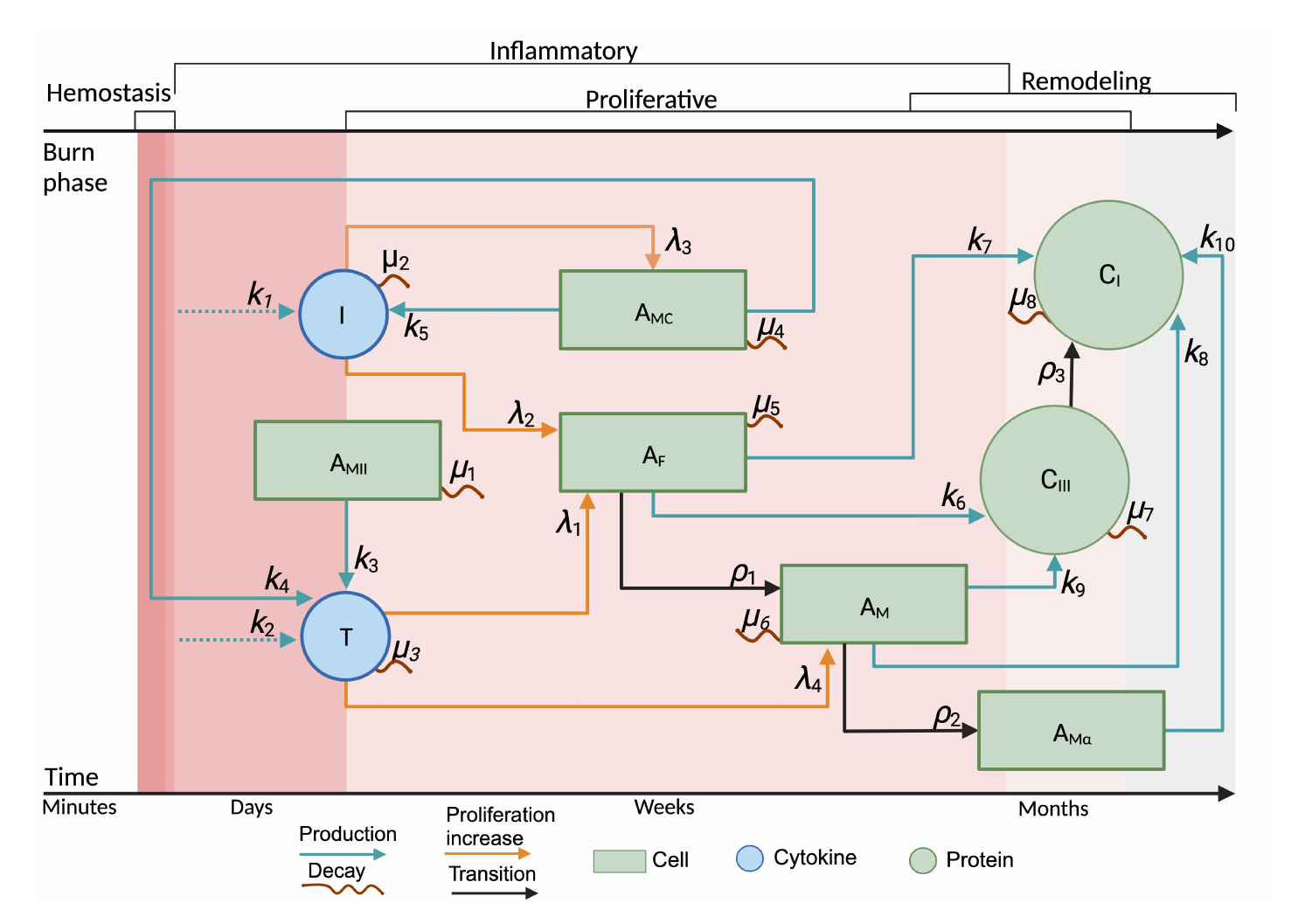
Cellular ODEs
The paper's concept of 'Cellular ODEs' represents a sophisticated computational approach to understanding burn wound healing. It utilizes a system of coupled ordinary differential equations (ODEs) to mathematically model the complex, dynamic interplay between various cell types, cytokines, and proteins over time. This framework is invaluable as it translates core biological processes - like cell proliferation, decay, and transition between phenotypes (e.g., fibroblasts to myofibroblasts) - into a quantitative, predictive model. By simulating the temporal evolution of these cellular and molecular components, the model allows researchers to test complex scenarios and the impact of signaling dynamics on healing outcomes. This powerful a priori computational method effectively bridges the gap between biological observation and systems-level understanding.
Signal Duality
The concept of 'Signal Duality' in this study refers to the complex and often conflicting interplay between pro-inflammatory (PI) and anti-inflammatory (AI) signals that govern the transition to the proliferative phase of wound healing. The paper's computational model cleverly explores this through two scenarios: one with dominant AI signals punctuated by PI spikes, and another featuring an ambiguous, intertwined oscillation of both signal types. This duality is critical, as the delicate balance between these opposing forces dictates the healing trajectory. The model reveals how sustained inflammation versus a regulated transition significantly impacts fibroblast activity and collagen deposition, ultimately determining whether healing is successful or impaired. This highlights that healing is governed not by simple signals but by their dynamic and context-dependent balance.
Virtual Wounds
The concept of 'Virtual Wounds' embodies this paper's core innovation: leveraging computational models to simulate the complex, non-linear dynamics of the burn healing process. This in-silico approach creates a digital replica of the wound's proliferative phase, enabling a deep exploration of the intricate interplay between key players like cytokines, fibroblasts, and collagen types. By testing different immunological scenarios, such as varying the influence of pro- and anti-inflammatory signals, the model reveals how subtle shifts in these initial conditions can dramatically alter healing trajectories, potentially leading to delayed repair or pathological scarring. This predictive capability moves beyond static observation, offering a dynamic framework to understand underlying biological mechanisms and paving the way for future personalized treatment strategies.
Predict & Treat
This research paper's computational model lays a foundational framework for a future "Predict & Treat" paradigm in burn wound care. The model's strength lies in its ability to predict the complex, non-linear trajectory of healing by simulating the dynamic interplay between key cellular and molecular components under different inflammatory scenarios. By inputting initial conditions, one could foresee potential pathological outcomes, such as excessive scarring or delayed remodeling. This predictive insight directly informs the "treat" aspect, enabling the *in silico* testing of targeted interventions. For instance, the model could simulate how modulating specific cytokines or cellular transition rates might steer the healing process towards a more favorable state, thus paving the way for developing personalized therapeutic strategies that are proactive rather than reactive.
More Figures (2)