Key Takeaways
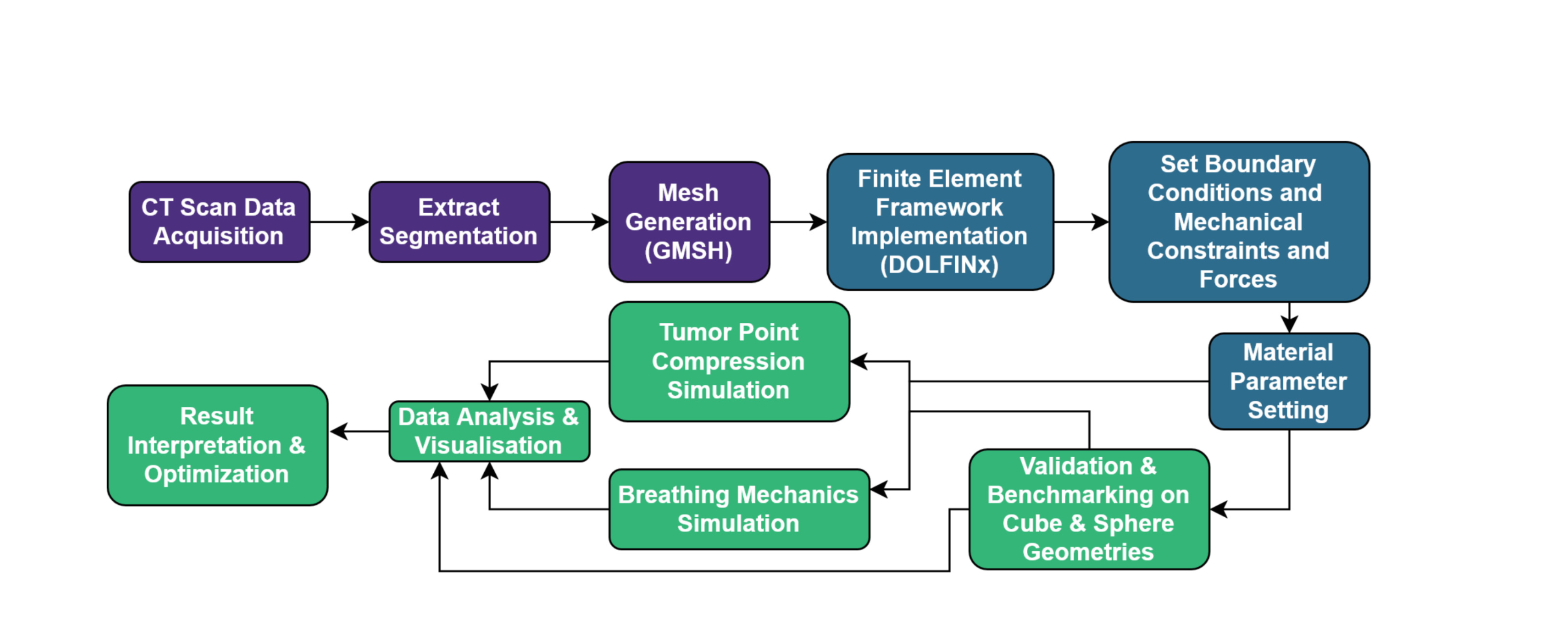
- The paper presents a validated computational framework using the Finite Element Method to model the complex biomechanical interactions between tumors and surrounding tissues, specifically for mesothelioma and the lung.
- The model successfully simulates realistic tissue deformation, including localized compression on a tumor and lung volume changes that mimic breathing cycles, using anatomical data from CT scans.
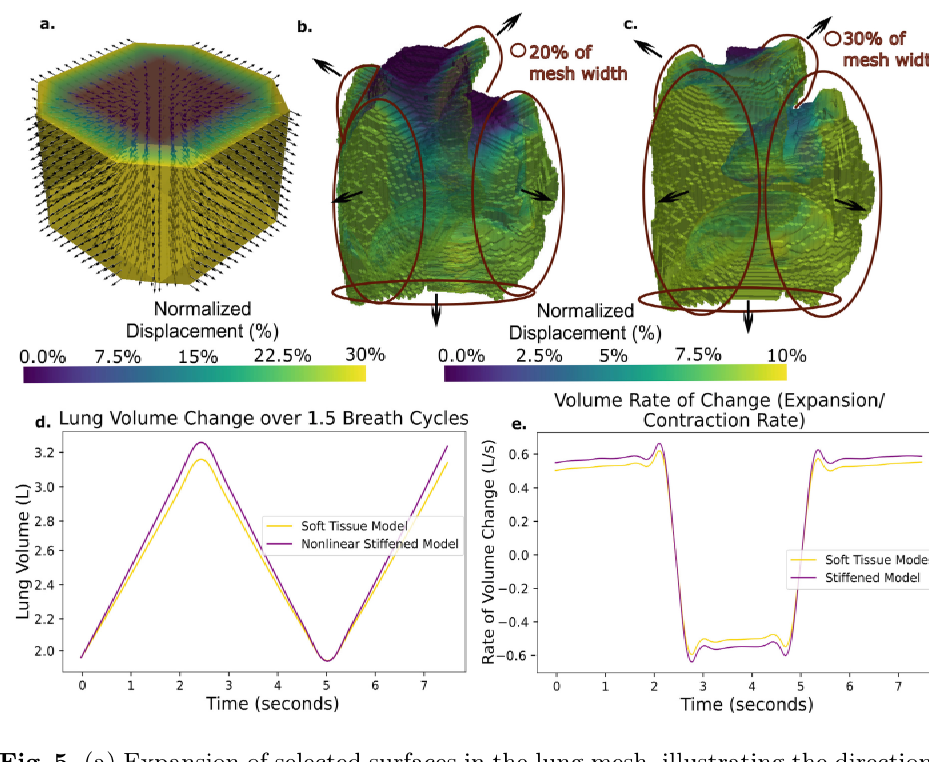
- The framework demonstrates how mechanical properties, such as increased tissue stiffness, directly impact deformation patterns and respiratory dynamics, highlighting their importance in tumor progression.
TL;DR
Cancers like malignant pleural mesothelioma (MPM) don't just grow in isolation; they physically push on and deform surrounding organs. The biomechanical forces from breathing and tissue stiffness play a crucial but often overlooked role in how these tumors grow and spread. Existing computational models frequently simplify these complex physical interactions, limiting our ability to accurately predict tumor behavior in a patient-specific way. This paper introduces a computational framework using the Finite Element Method (FEM) to realistically simulate these forces. Researchers created a 3D model of a lung and a synthetic tumor from actual CT scans. They successfully simulated how the tumor and lung deform under pressure and during breathing-like expansion. This proof-of-concept provides a verifiable foundation for building personalized models that can help predict tumor invasion and plan better treatments for MPM.
Why Does It Matter?
This paper provides a versatile and validated framework for integrating biomechanics into cancer modeling. It allows for the simulation of patient-specific tumor-tissue interactions from medical imaging, moving beyond overly simplified models. This is a crucial step towards creating predictive tools for tumor growth and invasion pathways, offering significant potential for developing personalized medicine strategies in oncology.
Biomech Context
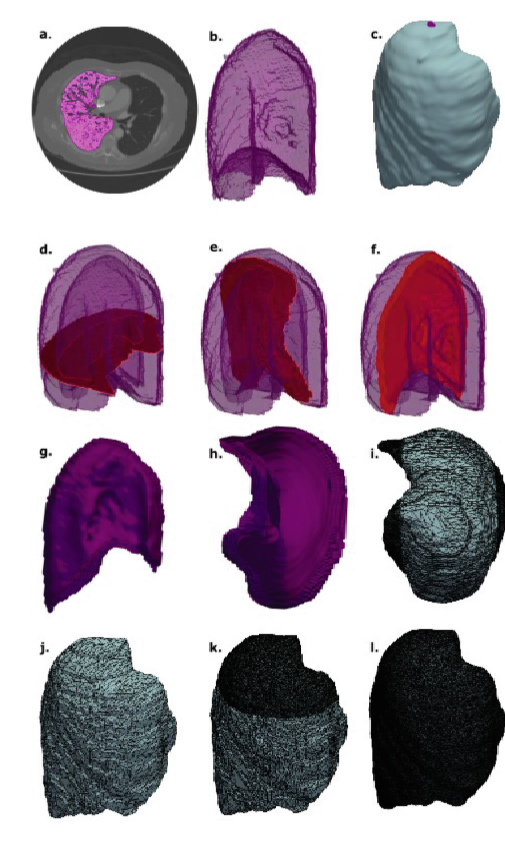
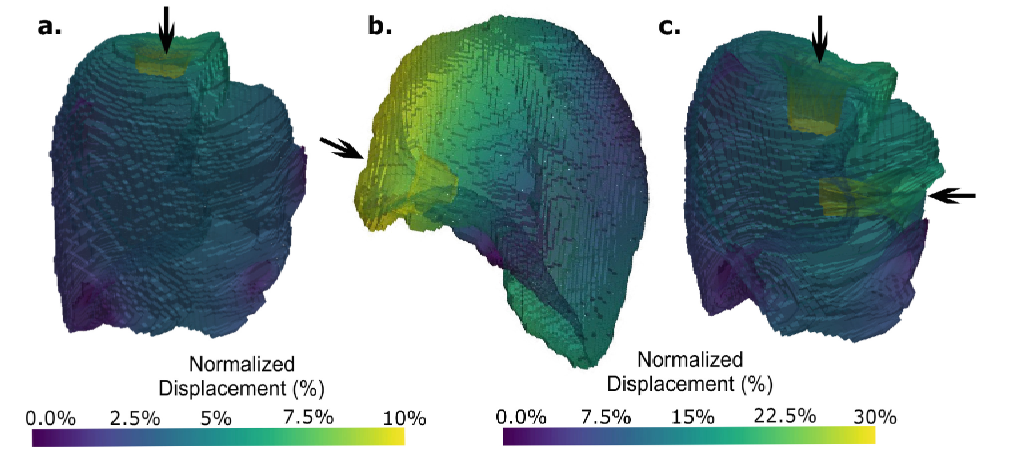
The biomechanical context of this research is profoundly significant, moving beyond traditional biological models to quantify the physical interplay between tumours and host tissues. The study establishes a computational framework that leverages patient-specific CT data to create realistic anatomical models, enabling the simulation of complex force dynamics in Malignant Pleural Mesothelioma. It critically highlights how solid stress and mechanical forces are not passive elements but active drivers of tumour progression and invasion. A key insight is the model's ability to simulate respiratory mechanics, demonstrating how increased tumour stiffness directly impedes lung expansion and alters breathing dynamics. This proof-of-concept provides a robust foundation for personalized medicine, offering a way to predict how a specific tumour will deform and interact with its environment.
In Silico Lung
The 'In Silico Lung' represents a powerful, patient-specific digital twin constructed from medical imaging. This isn't merely a static 3D model; it's a dynamic computational framework built using the Finite Element Method (FEM) to simulate complex biomechanics. The research demonstrates how this virtual lung, derived from CT scans, can replicate the physical stresses and deformations caused by a mesothelioma tumor. Crucially, the model simulates respiratory dynamics, capturing how lung tissue expands, contracts, and stiffens under strain, mirroring real physiological processes. By modeling the intricate force-play between the tumor and the lung, this approach provides a non-invasive platform to predict tumor growth pathways, assess the impact on breathing, and ultimately paves the way for personalized therapeutic strategies.
Fibrosis Mimic
The concept of a 'Fibrosis Mimic' is foundational to this computational framework, using material properties to simulate pathological tissue hardening. The study models the mesothelioma tumour with a significantly higher stiffness (Young's modulus) to emulate the mechanical characteristics of fibrotic and advanced tumours. Moreover, a 'stiffened lung model' is introduced specifically to represent the biomechanical effects of conditions like pulmonary fibrosis. This approach is insightful because it allows the simulation to capture how increased tissue stiffness, a hallmark of fibrosis, directly restricts lung expansion and alters breathing dynamics, aligning with clinical observations. By mimicking the strain-dependent stiffening of fibrotic tissue, the model uncovers the dynamic consequences of tumour growth on organ function.
Simple Physics
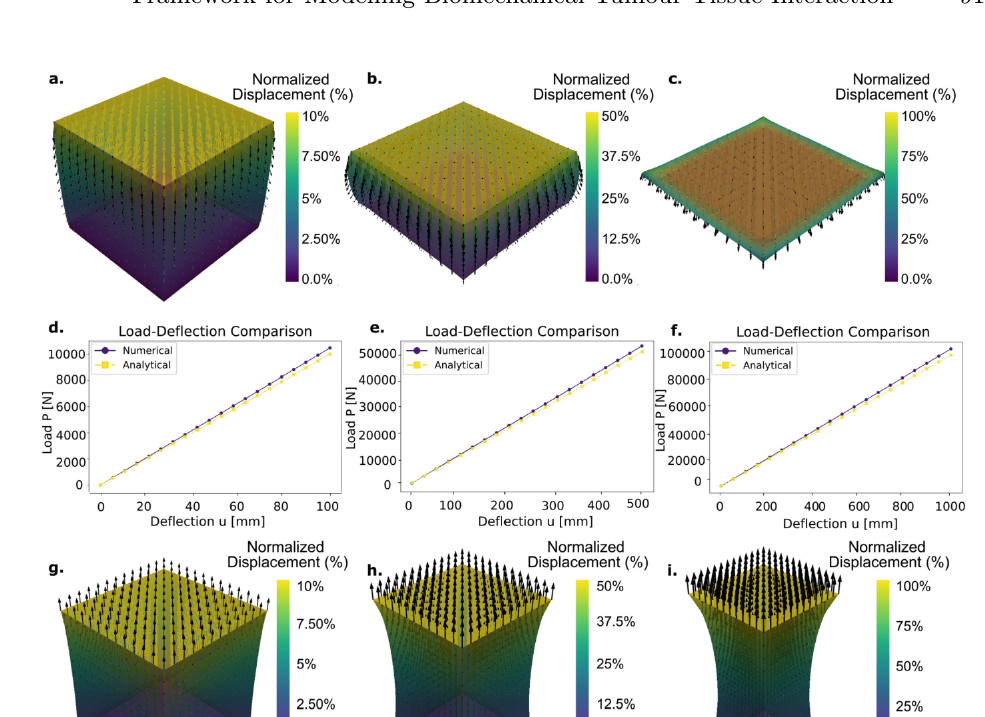
The 'Simple Physics' section of this research serves as the critical bedrock for the entire computational framework, demonstrating a deliberate and rigorous validation process. Before tackling the intricate biomechanics of a mesothelioma tumour within a lung, the authors strip the problem down to its core principles. By applying linear elasticity models to a simple cube geometry, they can directly compare their numerical simulation results against a known analytical solution derived from Hooke's Law. The exceptional agreement, with deviations under 5%, is not merely a technicality; it's a profound proof-of-concept that establishes the model's fundamental accuracy. This foundational step ensures confidence that the complex, patient-specific simulations built atop this framework are reliable, laying a robust foundation for predictive medicine.
Towards Growth
This research establishes a powerful computational framework for understanding biomechanics, but its true potential lies in the future developments implied by 'Towards Growth'. The next phase aims to transcend the current static model by simulating time-dependent tumor progression, allowing prediction of invasion pathways based on mechanical feedback from the surrounding environment. Growth also means enriching the model by integrating complex biological processes like nutrient transport and cellular dynamics, evolving it into a multi-scale, multi-physics tool. Ultimately, this trajectory points towards a significant clinical impact through predictive personalized medicine, where patient-specific simulations could forecast tumor development, guide surgical planning, and refine treatment for mesothelioma, transforming computational models into actionable clinical insights.
More Figures (4)
Stay Updated with Our Research
Get notified when we publish new AI-analyzed papers and research insights:
Subscribe to Newsletter