Key Takeaways
- A novel computational model was developed to simulate the progression of Type 2 Diabetes (T2D) by integrating five key biological hallmarks.
- The model demonstrates that chronic stress (CS) is a critical modulator that disrupts normal glucose-insulin dynamics and accelerates the progression towards a diabetic state.
- The study proposes a method to quantify a "Disease Progression" (DP) score, showing a direct link between the introduction of stress and an increase in the rate of T2D onset.
TL;DR
Type 2 Diabetes (T2D) is a growing global health issue where the body struggles to regulate blood sugar. While lifestyle factors are known contributors, the link between chronic stress (CS) and T2D is complex and not fully understood. Scientists know that stress affects hormones and metabolism, but it's been difficult to pinpoint exactly how this long-term wear and tear, or allostatic load, drives the progression from a healthy state to full-blown diabetes. This paper introduces a computational model that simulates the complex biological system involved in T2D. By combining several key biological markers, or hallmarks, of the disease (like insulin resistance and high glucose), the model can track disease progression (DP) over time. The key contribution is integrating the effects of chronic stress into this simulation, demonstrating quantitatively how stress disrupts the body's balance and acts as a critical driver, accelerating the onset of T2D.
Why Does It Matter?
This work provides a crucial mechanistic modeling framework for studying the complex link between chronic stress and T2D. It offers a quantitative tool to simulate disease progression and evaluate the impact of different stressors *in-silico*. This can accelerate research into disease mechanisms and potential therapeutic interventions by allowing for rapid hypothesis testing that would be difficult or unethical in clinical studies.
Allostatic Load
The concept of allostatic load is central to this study, signifying the cumulative physiological "wear and tear" on the body resulting from chronic stress. The paper's key insight is its use of this concept to operationalize chronic stress, transforming an abstract stressor into measurable physiological units. This is achieved by quantifying the load through five specific biological hallmarks linked to Type 2 Diabetes (T2D) progression: Insulin Resistance, Hyperglycemia, Low-Grade Inflammation, Hypercortisolism, and Hyperglucagonemia. The computational model calculates a "Disease Progression" (DP) percentage based on the cumulative strain across these hallmarks, thus creating a powerful quantitative bridge between the experience of stress and the physiological onset and progression of T2D disease.
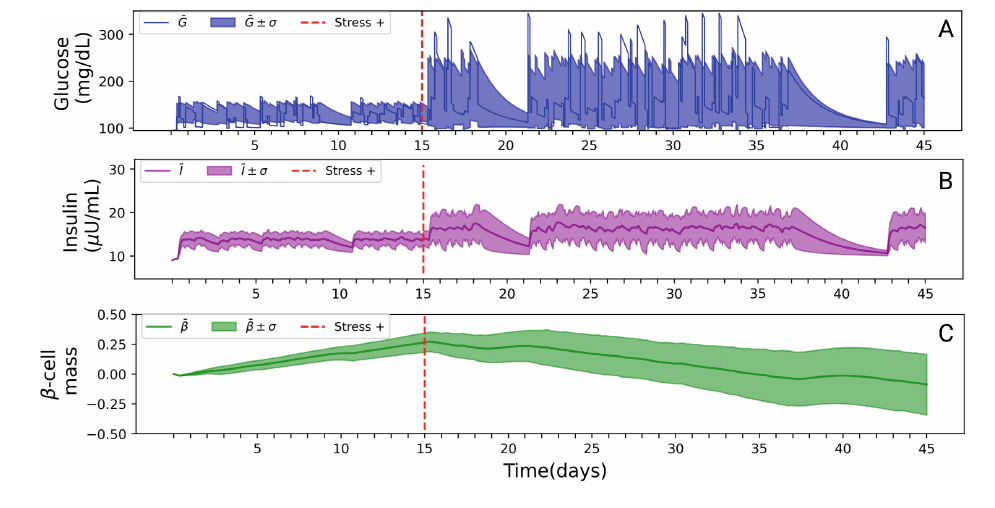
In-Silico Stress
"In-silico stress," as conceptualized in this research, is a digital proxy for the physiological effects of chronic stress (CS) on human metabolism. It involves applying quantifiable disturbances to a computational model that simulates the complex interplay between glucose, insulin, and β-cells. By altering parameters to mimic the aberrant glucose spikes and metabolic strain characteristic of a stressed individual, the model can predict disease progression. This approach provides a crucial advantage: it isolates stress as an experimental variable, allowing researchers to observe its direct causal chain of effects - from initial hormonal disruption to the eventual development of Type 2 Diabetes hallmarks like hyperglycemia. Ultimately, this *in-silico* methodology quantifies the "allostatic load" of stress and creates a powerful tool for exploring the mechanisms of T2D onset in a controlled, virtual setting.
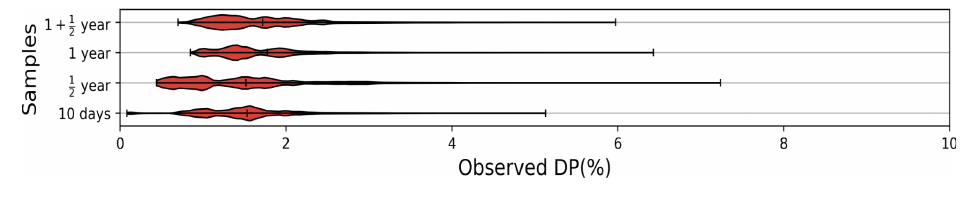
Quantifying DP
The paper’s approach to “Quantifying DP” (Disease Progression) provides a sophisticated computational framework for translating physiological strain into a tangible, measurable metric. Instead of relying on a single biomarker, this method creates a holistic “damage” score by monitoring multiple key biological hallmarks of Type 2 Diabetes, such as hyperglycemia and insulin resistance, under simulated chronic stress. The model defines pathological thresholds for each hallmark, and when a simulated value crosses a threshold, it registers a “strain event.” The final DP is ingeniously calculated as a cumulative percentage, representing the aggregated “allostatic load” or total “wear and tear” from all strain events across all hallmarks over time. This provides a unified, dynamic assessment of disease advancement.
Model Limits
A significant limitation of the presented model is its reliance on in-silico simulated data, positioning it as a conceptual proof-of-concept rather than a clinically validated tool. This computational approach, while demonstrating the interplay between chronic stress and Type 2 Diabetes (T2D), inherently lacks the complexity and variability of real-world patient physiology. The model is built upon a simplified minimal framework for glucose-insulin dynamics, and it abstracts certain factors, such as the casual inclusion of Low Grade Inflammation (LGI), which would require more direct clinical measurements for true accuracy. The study also notes ambiguities regarding the optimal simulation time-span. Therefore, the model's true potential is contingent upon future validation against empirical, real-life data to refine its parameters and confirm its predictive power.
Need Real Data
The paper's computational model, while demonstrating plausible *in-silico* dynamics between chronic stress and Type 2 Diabetes, fundamentally underscores the imperative need for real-world data. The authors explicitly concede that their simulations, built upon existing literature and theoretical constructs, provide only "limited information." This highlights a critical gap: the model's current inability to capture the nuanced, chaotic realities of individual patient physiology and disease progression. To transition from a promising conceptual framework to a clinically relevant tool, empirical validation is non-negotiable. Real patient data is essential not only to calibrate and refine the model's parameters but also to test its predictive power against the diverse manifestations of stress and T2D, thereby moving beyond a sophisticated but theoretical hypothesis.
More Figures (1)