Key Takeaways
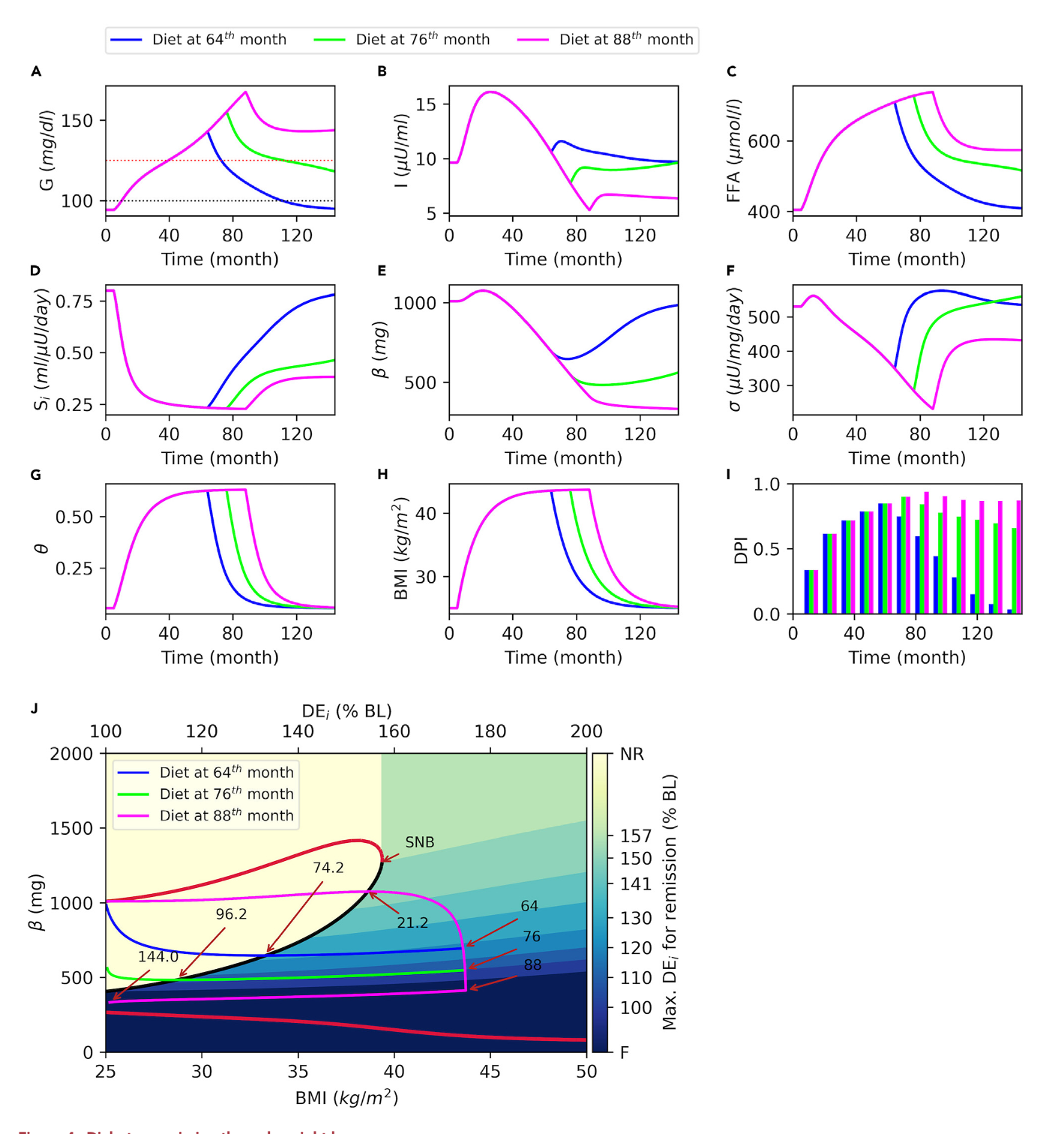
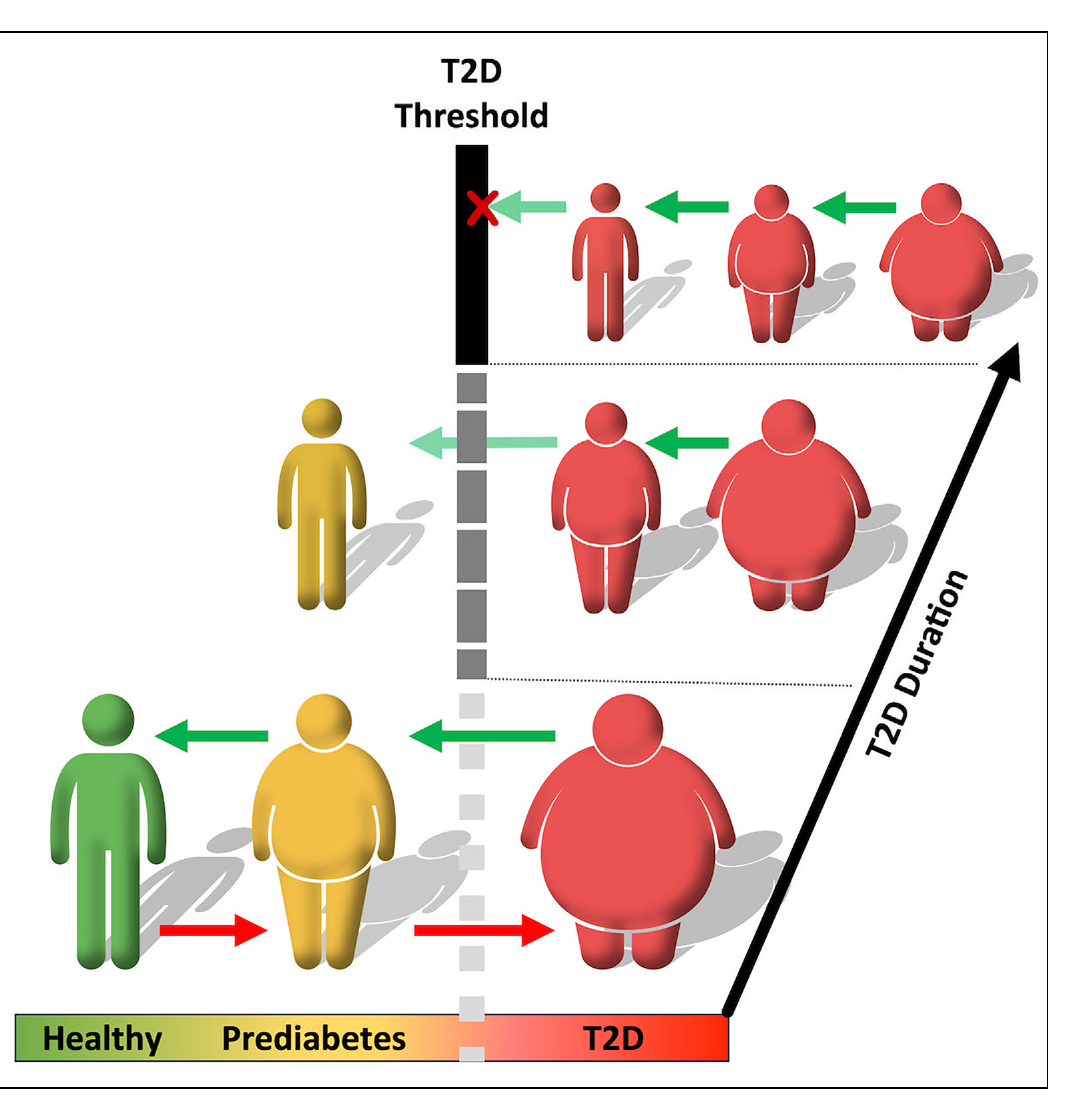
- The model identifies a critical “window of opportunity” for T2D remission; the longer an individual has had diabetes, the more significant the calorie restriction required to reverse it.
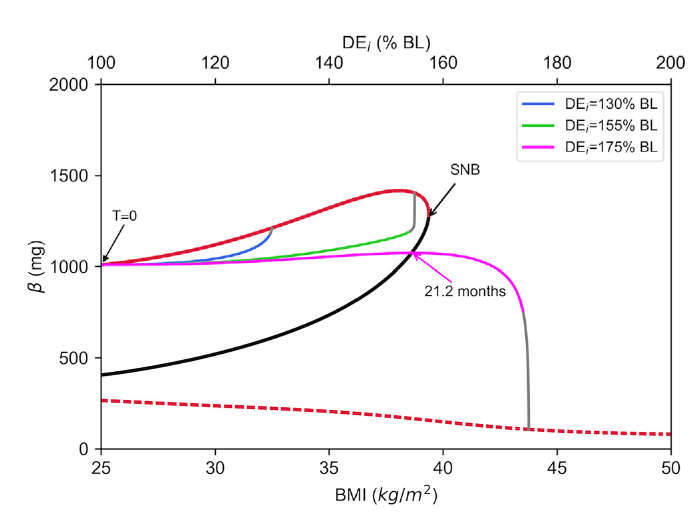
- A decline in pancreatic β-cell function starts well before the clinical diagnosis of T2D, and the remaining functional β-cell mass is a key predictor of whether remission is possible.
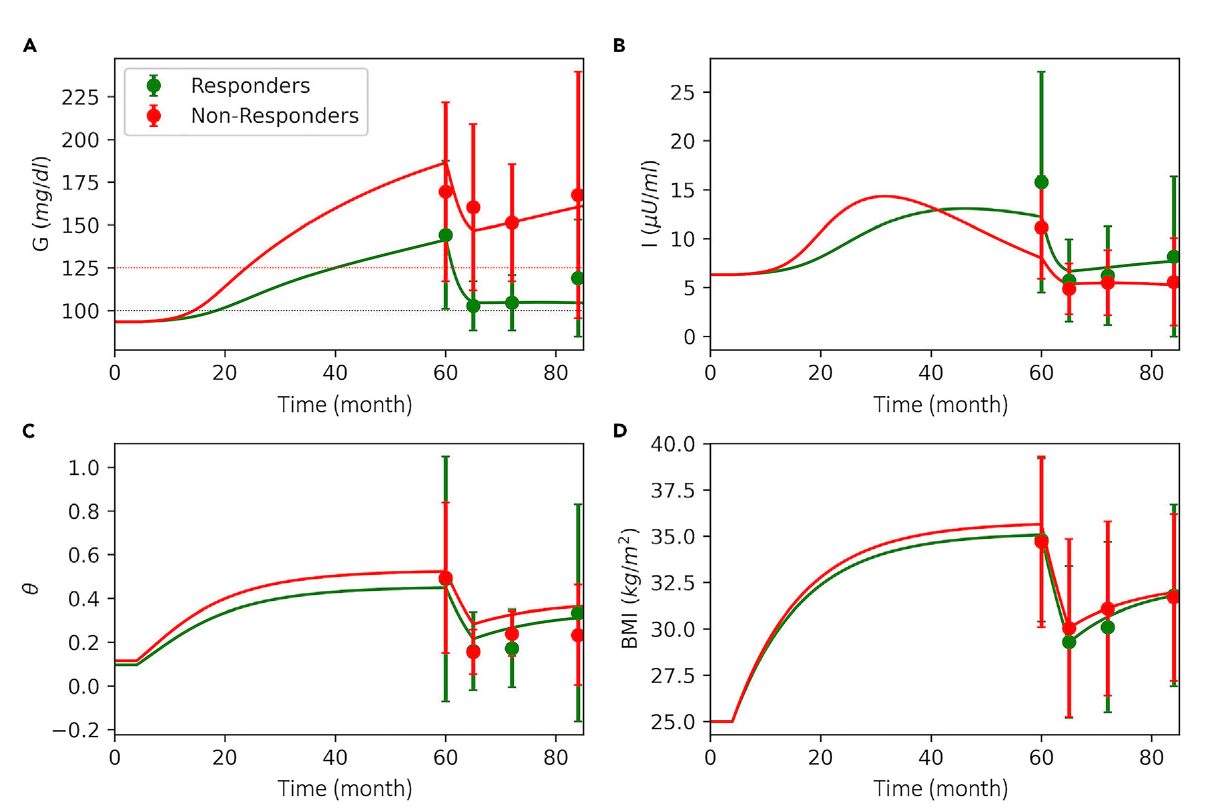
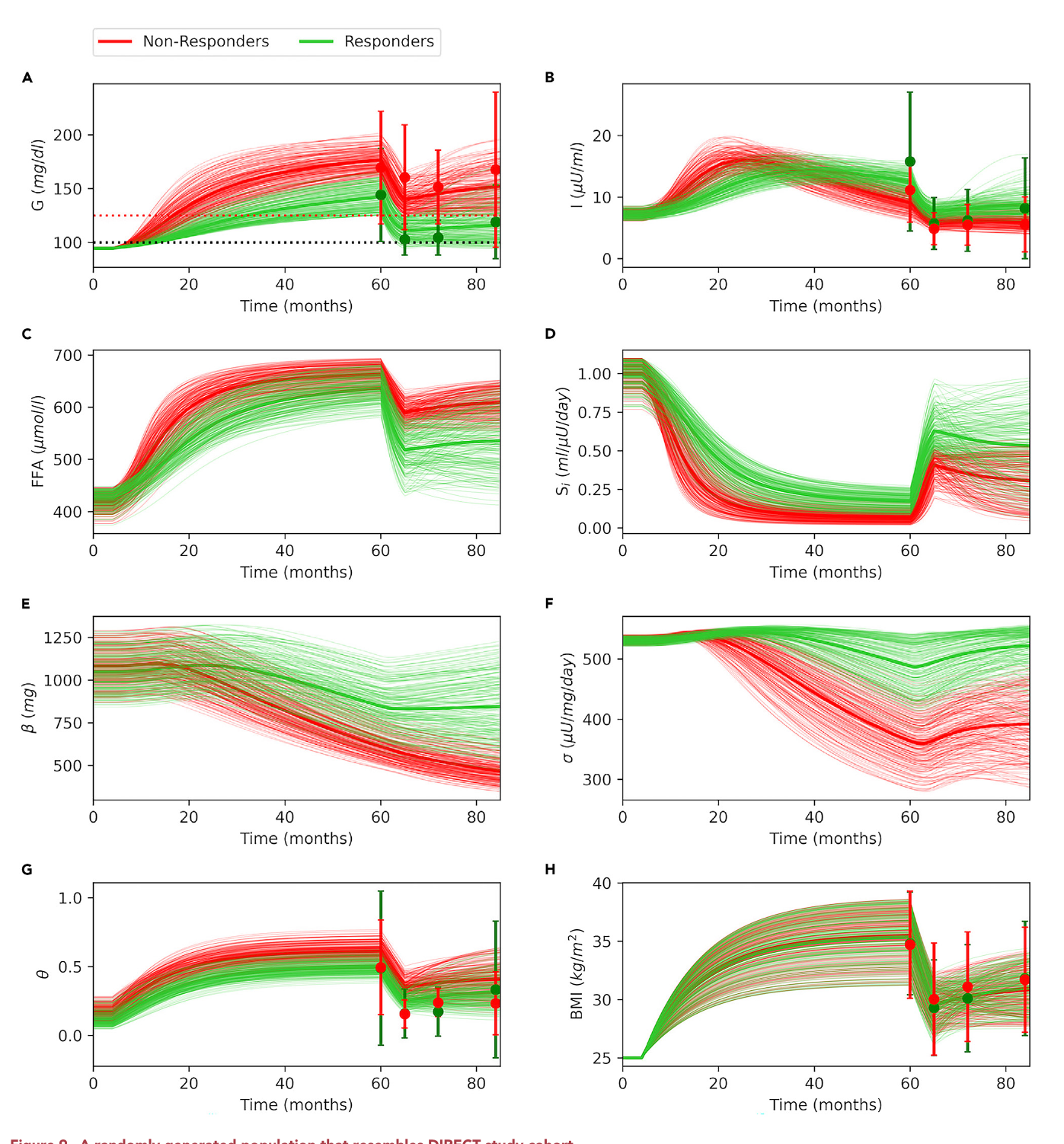
- The model successfully replicated the results of the DiRECT clinical trial, providing a mechanistic explanation for why some individuals achieve remission through weight loss (“responders”) while others do not.
TL;DR
Obesity is a major risk factor for type 2 diabetes (T2D). While significant weight loss can lead to remission, it's not a guaranteed outcome, and the specific factors that determine success remain unclear. Existing studies often can't track the complex, long-term interactions between weight, insulin production, and other metabolic variables. This knowledge gap makes it challenging to predict who will benefit from weight-loss interventions and to optimize treatment strategies for individuals.
Why Does It Matter?
This paper offers a powerful mechanistic model that integrates key physiological factors in T2D progression and remission. It provides a computational framework to test hypotheses virtually, analyze long-term intervention outcomes, and understand patient variability. This can help personalize treatment strategies by identifying individuals most likely to benefit from weight-loss interventions, bridging the gap between clinical data and underlying biological processes.
In Silico Lab
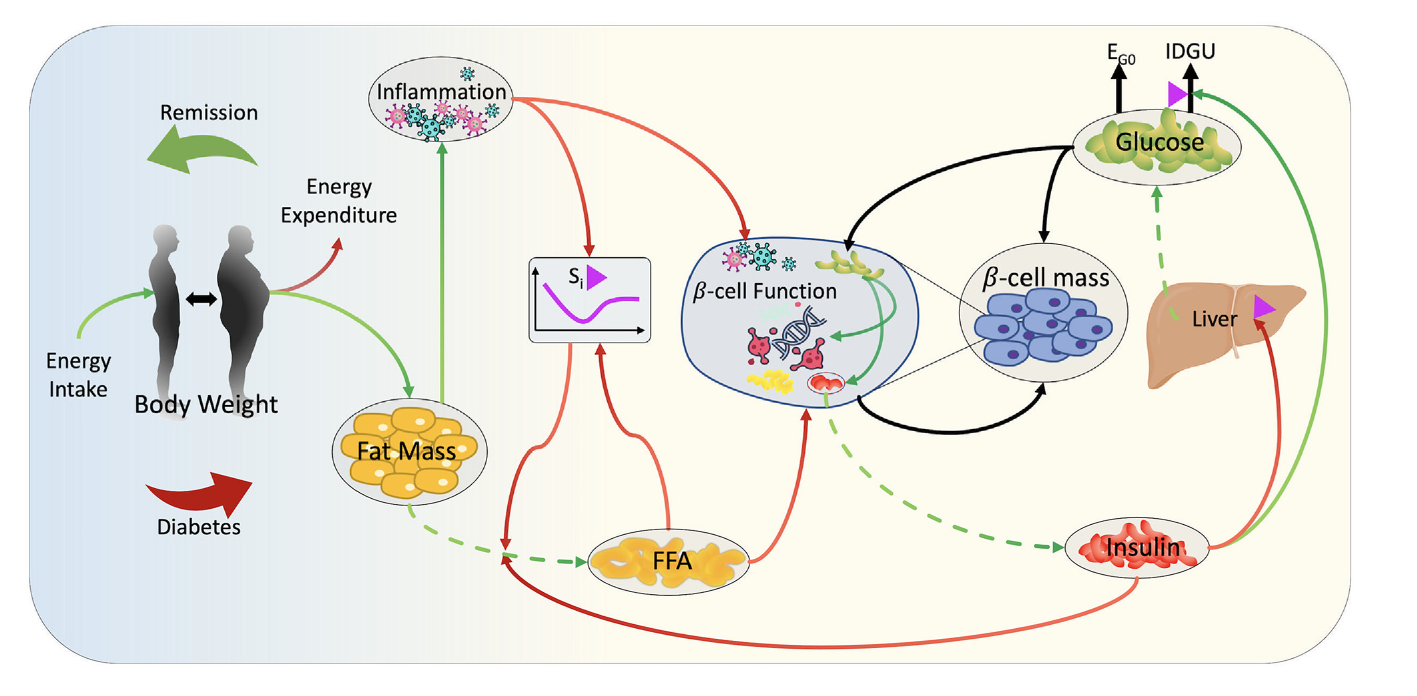
The 'In Silico Lab' represents a powerful computational replica of human metabolism, designed to explore the dynamics of obesity-driven diabetes. This virtual environment elegantly models the intricate feedback loops connecting glucose, insulin, systemic inflammation, and beta-cell function, providing a holistic system view. Its core strength lies in simulating long-term "what-if" scenarios over years, a feat impractical in human trials. Through virtual experiments, such as manipulating dietary intake, the model pinpoints critical mechanistic thresholds and "windows of opportunity" for disease remission. By generating diverse virtual patient populations, it can reproduce real-world clinical trial outcomes, explaining individual variability in remission success and offering a predictive tool to optimize therapeutic strategies.
Tipping Points
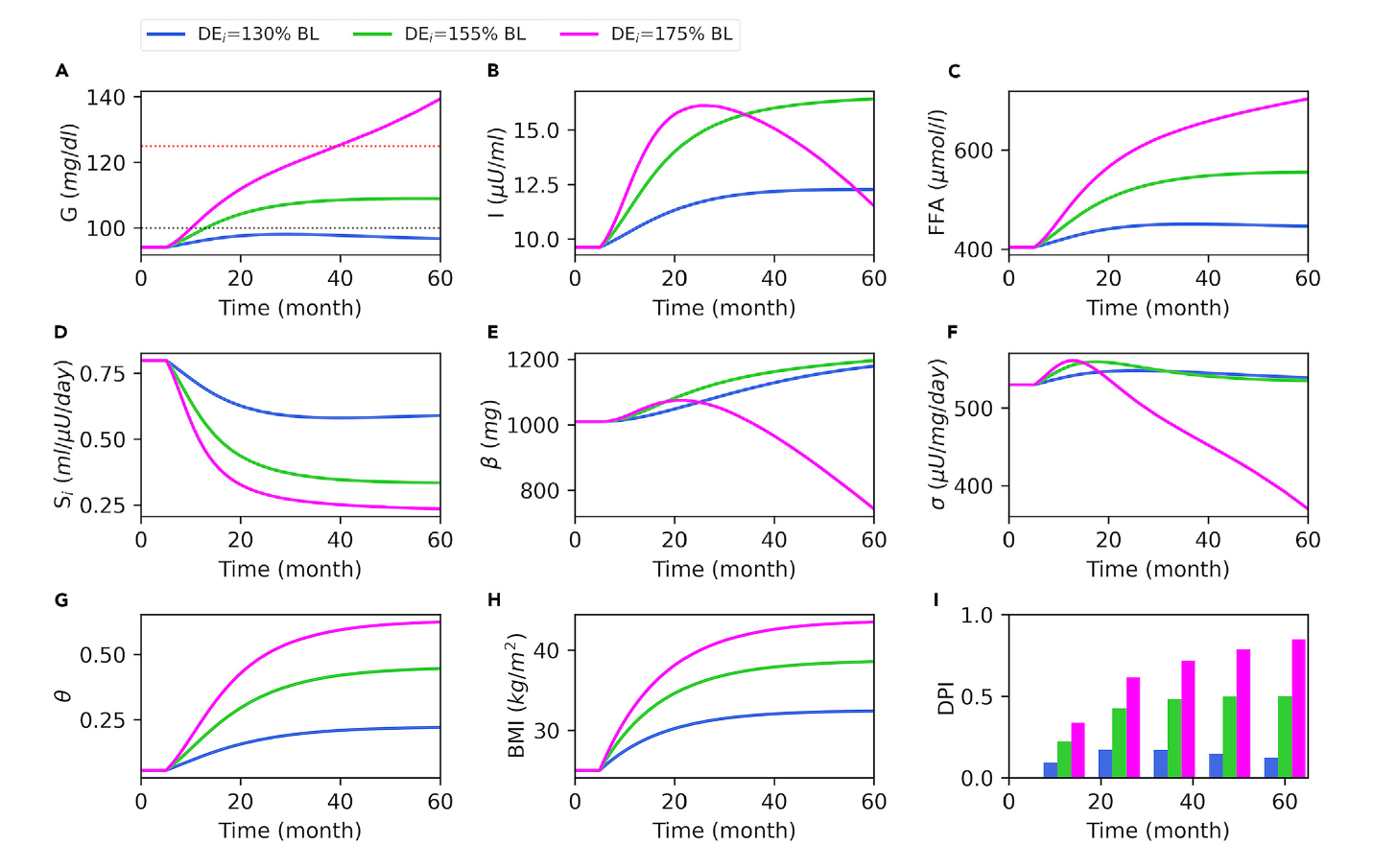
This paper's model reveals that the onset of Type 2 Diabetes (T2D) is not gradual but involves a critical tipping point. This threshold, governed by factors like BMI and declining β-cell function, represents an abrupt shift from a stable healthy/prediabetic state to a stable diabetic state, a phenomenon known as bistability. Crossing this point is not easily reversible. The model uncovers a crucial, time-sensitive window of opportunity for remission through weight loss. The longer an individual has had T2D and the further they are past this tipping point, the more difficult remission becomes. The analysis underscores that the system's "memory" of the diabetic state makes early and aggressive intervention vital for successful reversal, as the path back is not the same as the path to onset.
Virtual Cohort
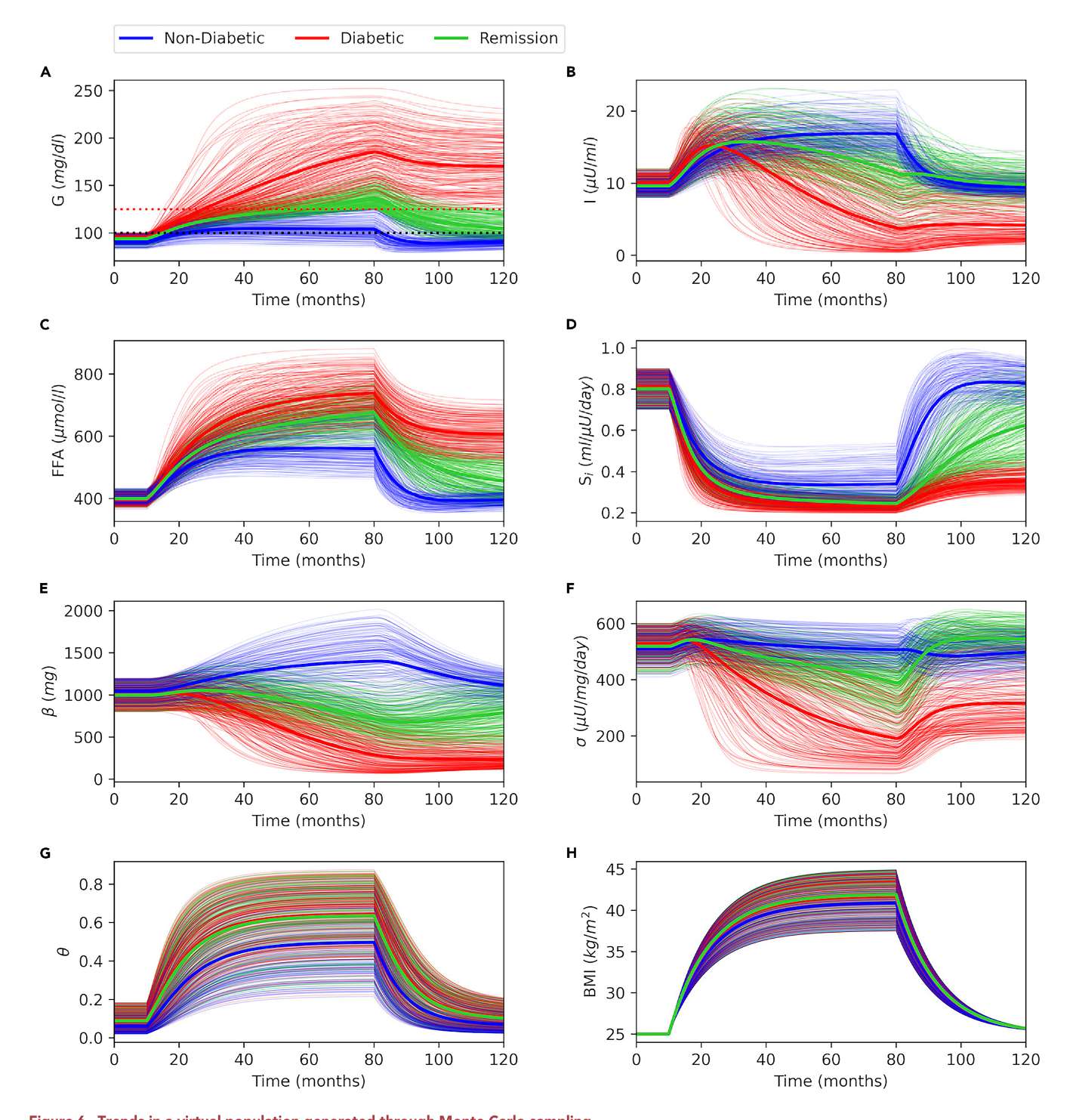
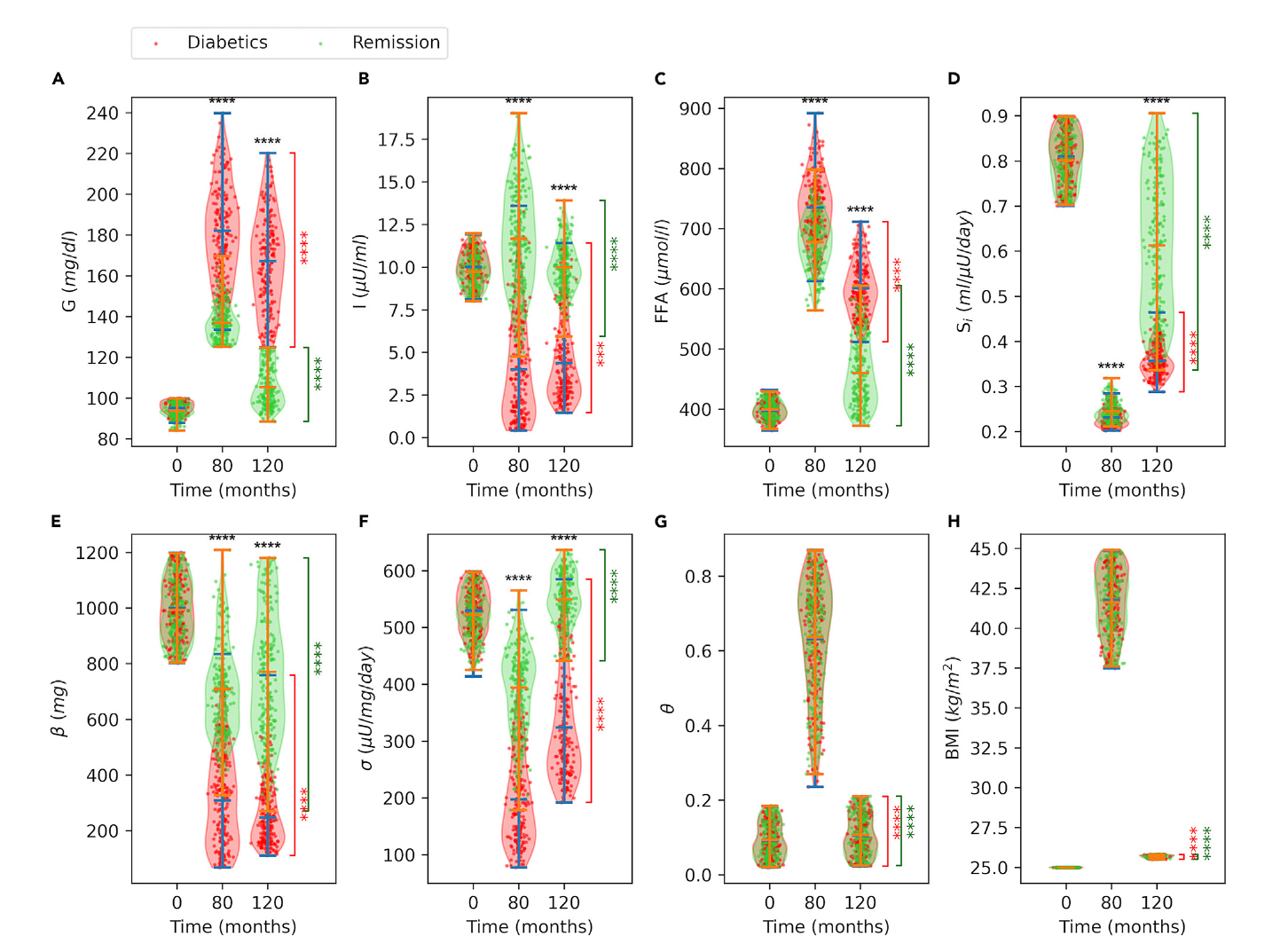
The 'virtual cohort' is a powerful computational tool used to simulate population heterogeneity in diabetes. Instead of a single patient model, it employs Monte Carlo sampling to generate a large population of unique 'virtual individuals' with varied metabolic parameters. This method allows for a nuanced exploration of the diverse physiological trajectories leading to diabetes onset and remission. A key strength is the model's ability to generate a virtual cohort that reproduces trends from a real-world clinical trial (the DIRECT study). By simulating and comparing 'responders' and 'non-responders' to a weight-loss intervention, the study effectively uncovers the critical underlying factors, like β-cell function, that distinguish these groups, offering deep insights into personalized medicine.
Inflamm. Role
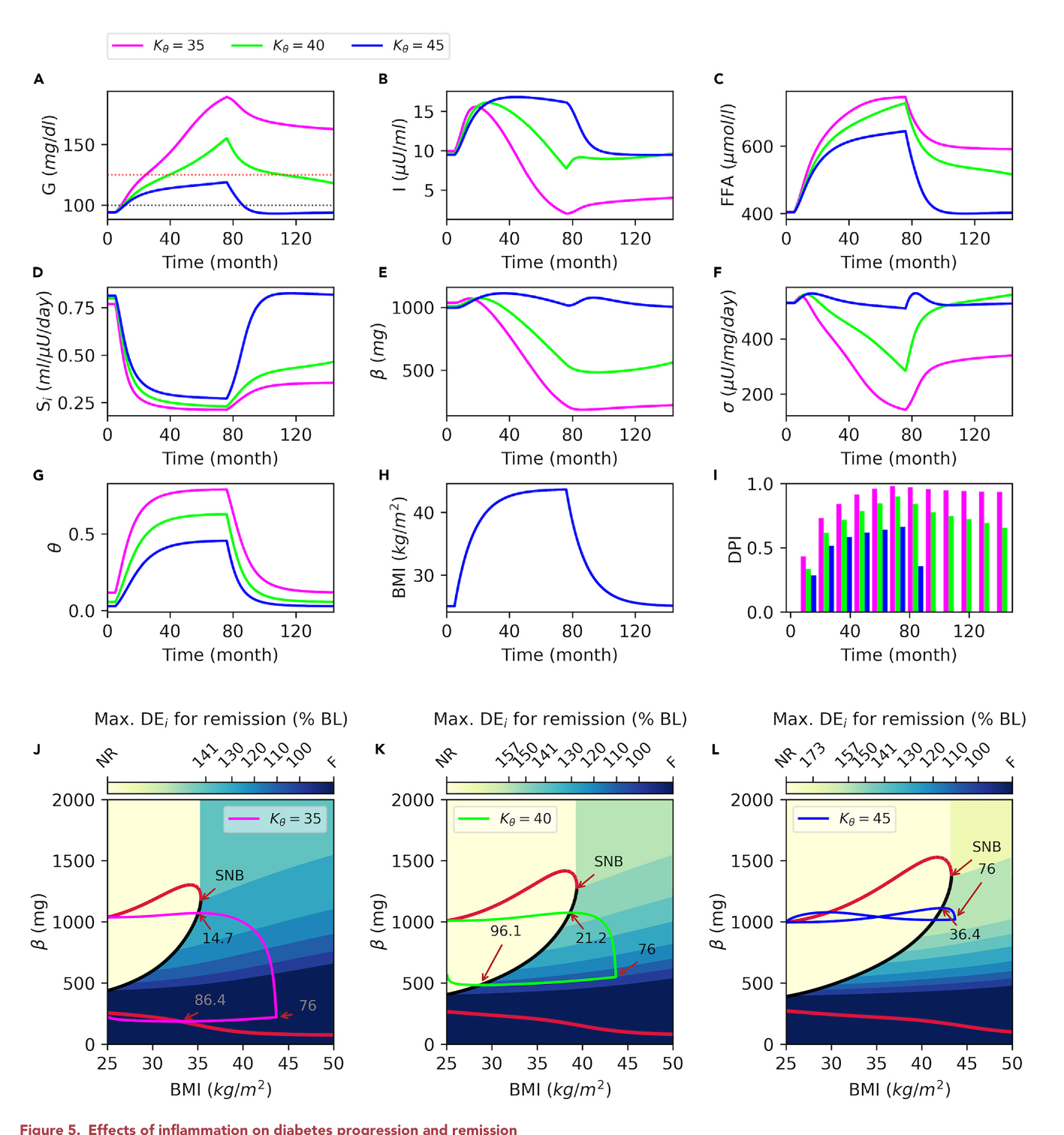
This computational model powerfully illustrates that inflammation acts as a critical amplifier in the progression of obesity-driven Type 2 Diabetes (T2D). The analysis reveals that an individual's inherent inflammatory response to weight gain significantly dictates their disease trajectory. A stronger inflammatory reaction not only accelerates the onset of T2D by more rapidly degrading insulin sensitivity and β-cell function, but it also critically hampers the success of remission. The model demonstrates that heightened inflammation necessitates a far more drastic and sustained caloric restriction to reverse the diabetic state. This effectively shrinks the 'window of opportunity' for remission, making recovery much more challenging and highlighting inflammation as a key factor in determining both an individual's susceptibility and their potential to recover from T2D.
Beyond Fasting
This computational model transcends a focus on simple fasting states, framing Type 2 Diabetes as a dynamic process. The analysis reveals that successful remission through weight loss is not guaranteed but depends on a critical "window of opportunity". This window is defined by the disease's duration and the patient's underlying metabolic state, including β-cell function and systemic inflammation. The model shows that individual trajectories diverge, meaning intervention success is highly personalized and time-sensitive. It posits that crossing specific dynamic thresholds can make remission significantly harder, demonstrating that reversing T2D requires a holistic understanding of the system's history and state, far beyond a single fasting measurement.
More Figures (9)